Table of contents
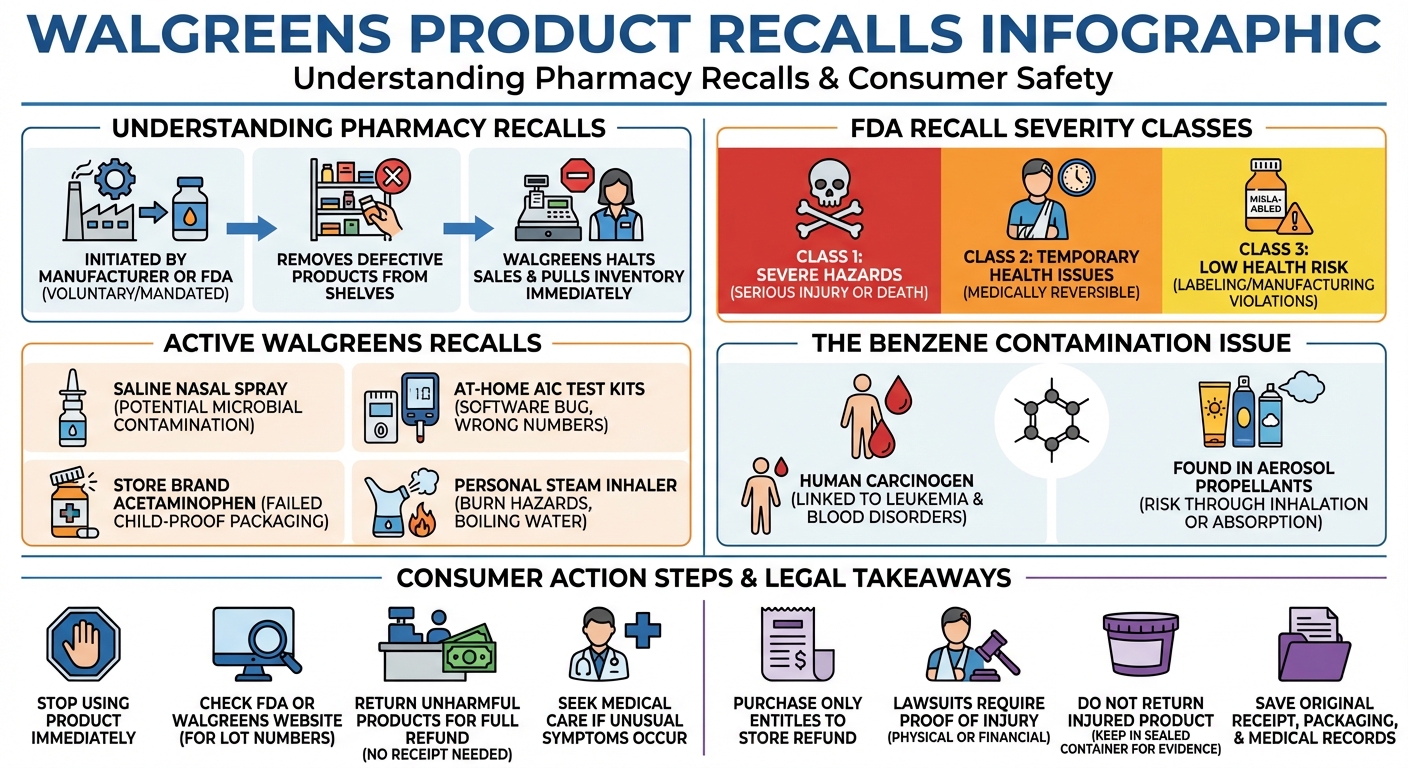
A Walgreens recall occurs when the FDA or a manufacturer removes defective or contaminated products from store shelves. Recent Walgreens recalls include the Saline Nasal Spray with Xylitol for bacterial contamination, At-Home A1C Test Kits for software bugs, and store-brand acetaminophen for defective child-resistant packaging.
Walgreens Product Recalls: Current Alerts and Consumer Rights
When you purchase over-the-counter medications, medical devices, or personal care items from a trusted pharmacy, you expect them to be safe. Unfortunately, manufacturing defects, software bugs, and chemical contaminations can turn everyday items into serious health hazards. Staying updated on Walgreens store-brand recalls is critical for protecting your health and the well-being of your family.
This guide provides a comprehensive overview of recent Walgreens product recalls, explains FDA recall classifications, and outlines your legal rights as a consumer. Use this information to check your medicine cabinet, safely dispose of dangerous products, and understand when a defective product might warrant a product liability claim.
Active Walgreens Recalls (2024-2025)
What nasal spray is being recalled?
The FDA recently issued a Class II recall for over 41,000 bottles of Walgreens Saline Nasal Spray with Xylitol. This recall was initiated due to potential microbial contamination. Consumers who purchased the 1.5-ounce bottles should stop using them immediately and return them to Walgreens for a full refund.
Walgreens At-Home A1C Test Kit Recall (Software Bug)
The FDA announced a Class 2 device recall for the Walgreens At-Home A1C Test Kit due to a critical software bug. The defect causes the device to display incorrect numerical values (less than 4% or greater than 13%) instead of the expected error messages. This malfunction can lead to inaccurate diabetes management and potentially dangerous medical decisions.
Aurohealth Walgreens Brand Acetaminophen (Packaging Defect)
Aurohealth recalled specific batches of Walgreens brand acetaminophen because the products failed to meet child-resistant packaging requirements mandated by the Poison Prevention Packaging Act. This defect poses a severe risk of accidental poisoning if young children gain access to the medication.
Well at Walgreens Personal Steam Inhaler (Burn Hazard)
The Well at Walgreens Personal Steam Inhaler was recalled after reports indicated that the device could produce boiling water and excessively hot steam during use. This design flaw presents a significant burn hazard to consumers, particularly when the device is used at a close distance to the face.
Understanding Pharmacy Recalls and FDA Guidelines
What is a product recall in pharmacy?
A product recall in a pharmacy is a voluntary or FDA-mandated action to remove defective or potentially harmful medications, devices, or consumer goods from the market. Recalls occur when a product violates FDA regulations, such as bacterial contamination, incorrect labeling, or manufacturing defects that pose health risks.
FDA Recall Classes Explained (Class I, II, and III)
The FDA categorizes recalls into three classes based on the severity of the hazard:
- Class I: The most serious type of recall. Use of the product will cause serious adverse health consequences or death.
- Class II: Use of the product may cause temporary or medically reversible adverse health consequences, but the probability of serious harm is remote.
- Class III: Use of the product is not likely to cause adverse health consequences, but it violates FDA labeling or manufacturing laws.
How Walgreens handles pulling contaminated products from shelves
When a recall is issued, Walgreens utilizes an internal centralized system to immediately halt the sale of the affected item at the register. Store managers are instructed to pull the inventory from shelves and either destroy it or return it to the manufacturer. Walgreens also posts recall notices on its official website and in physical store locations to alert consumers.
Chemical Contamination: The Benzene Issue
What products are being recalled for benzene?
Several aerosol-based products sold at pharmacies have been recalled for benzene contamination, a known human carcinogen. Recent recalls include specific batches of aerosol sunscreens, dry shampoos, antiperspirants, and antifungal sprays. Manufacturers voluntarily recalled these items after testing revealed trace amounts of benzene stemming from the aerosol propellants.
The risk of aerosol sprays, sunscreens, and dry shampoos sold in pharmacies
Benzene is not an intended ingredient in these personal care products. Instead, it is a byproduct of the manufacturing process, specifically related to the propellants used to spray the product out of the can. Because these products are applied directly to the skin or hair, the chemical can be absorbed into the bloodstream or inhaled.
Health risks associated with benzene exposure
Long-term exposure to benzene is highly dangerous. The chemical is known to cause cancer in humans, specifically leukemia and other blood disorders like aplastic anemia. Even trace amounts in daily-use products have prompted immediate FDA intervention and sweeping national recalls.
Legal Actions and Product Liability
What is the lawsuit against Walgreens pharmacy?
There is no single lawsuit against Walgreens; rather, the pharmacy faces multiple legal actions. These include a massive nationwide settlement regarding the dispensing of prescription opioids, as well as individual product liability lawsuits concerning defective store-brand products, contaminated medications, and failure to warn consumers about potential health hazards.
Distinguishing between mass torts and individual product liability claims
It is important to understand the difference between broad litigation and individual claims:
- Mass Torts: Large-scale litigation, such as the opioid settlement, where thousands of plaintiffs consolidate their claims against a corporation for widespread harm.
- Individual Product Liability: A lawsuit filed by a single consumer who suffered a specific, documented injury due to a defective product, such as a severe burn from a recalled steam inhaler.
When a product recall becomes a personal injury lawsuit
A product recall alone does not automatically entitle you to a lawsuit. To file a successful personal injury or product liability claim, you must prove that you actually used the defective product and suffered a measurable physical or financial injury as a direct result. If you simply purchased the item but were not harmed, your remedy is typically limited to a store refund.
What to Do If You Have a Recalled Walgreens Product
Steps for safely disposing of or returning recalled items for a refund
If you discover a recalled product in your home, stop using it immediately. Check the FDA website or the Walgreens recall page to confirm the lot number and UPC. Most recalls allow you to return the product to any Walgreens location for a full refund, even without a receipt. Do not pour contaminated medications down the drain unless specifically instructed to do so.
When to seek medical attention for adverse reactions
If you experience unusual symptoms, infections, or allergic reactions after using a recalled product—such as respiratory issues from a contaminated nasal spray or skin irritation from a benzene-tainted aerosol—seek medical attention immediately. Inform your doctor about the recalled product so they can accurately diagnose and treat the issue.
How to preserve evidence if you plan to consult a product liability attorney
If a recalled Walgreens product has caused you serious injury, do not return it to the store for a refund. Returning the item destroys your primary piece of evidence. Instead, place the product in a safe, sealed container. Keep your original receipt, the packaging, and all medical records related to your treatment. Then, consult with a qualified product liability attorney to discuss your legal options.