Table of contents
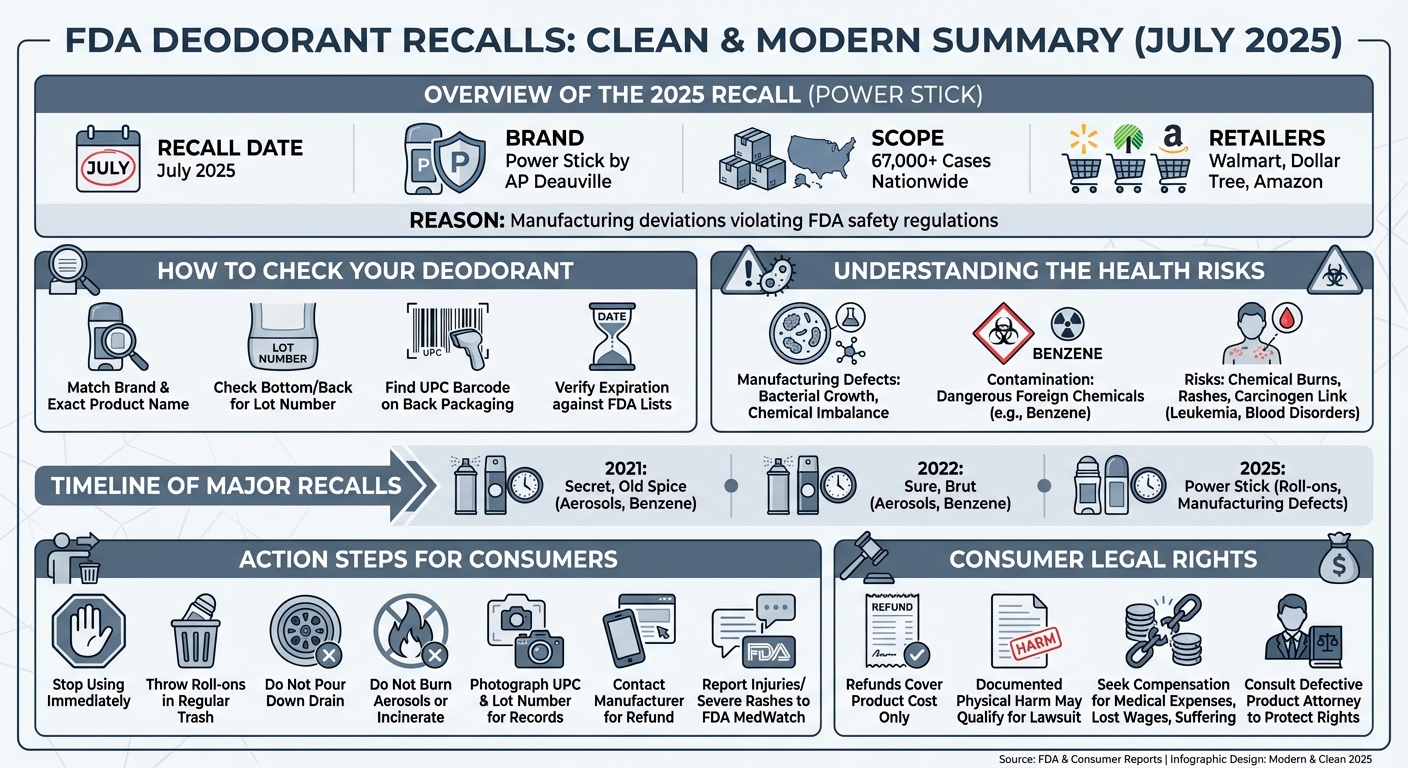
In July 2025, the FDA announced a nationwide recall of over 67,000 cases of Power Stick brand roll-on deodorants. The recall, initiated by manufacturer AP Deauville, was due to manufacturing deviations that violated FDA safety regulations. Consumers should stop using affected 1.8-ounce roll-on products immediately and request a refund.
FDA Deodorant Recall: What Consumers Need to Know
When you purchase personal care products, you expect them to be safe for daily use. However, recent FDA deodorant recalls have highlighted significant safety concerns, ranging from manufacturing defects to contamination with cancer-causing chemicals. Staying informed about these recalls is critical for protecting your health and understanding your legal rights as a consumer.
Recent FDA Actions on Antiperspirants and Deodorants
The U.S. Food and Drug Administration (FDA) closely monitors cosmetics and over-the-counter drugs, including antiperspirants. Over the past few years, the agency has overseen multiple nationwide recalls. These actions are typically triggered by independent laboratory testing, consumer complaints, or internal manufacturer audits that reveal deviations from strict safety regulations.
How to Identify if Your Product is Affected
If you suspect your deodorant is part of a recall, do not use it until you verify its safety. You can identify affected products by checking the following details on the packaging:
- Brand and Product Name: Ensure it matches the exact variety listed in the recall.
- Lot Number: Usually printed on the bottom of the canister or the back label.
- UPC Code: The barcode number on the back of the product.
- Expiration Date: Many recalls target specific manufacturing windows.
The 2025 Power Stick Deodorant Recall
In the summer of 2025, a massive recall disrupted the personal care market, affecting products sold at major retailers like Walmart, Dollar Tree, and Amazon.
Which deodorant has been recalled?
In July 2025, the FDA announced the nationwide recall of over 67,000 cases of Power Stick brand roll-on deodorants, manufactured by AP Deauville. Additionally, previous major recalls have included popular aerosol antiperspirants from brands like Secret, Old Spice, Sure, and Brut due to dangerous benzene contamination.
Why is Power Stick deodorant being recalled?
Power Stick deodorant is being recalled due to significant manufacturing deviations from FDA safety regulations. These quality control failures raised immediate safety concerns, prompting the manufacturer to pull over 67,000 cases of 1.8-ounce roll-on varieties from major retailers like Walmart, Dollar Tree, and Amazon.
List of Affected Power Stick Lot Numbers and UPCs
The July 2025 recall specifically targets three varieties of the 1.8-ounce Power Stick roll-on deodorant. Consumers are urged to check the bottom of their deodorant containers for the specific lot numbers listed on the FDA’s official recall page. If your product’s lot number matches the FDA’s list, it is part of the defective batch.
Historical Deodorant Recalls and Safety Concerns
The 2025 Power Stick recall is not an isolated incident. The personal care industry has faced several high-profile recalls in recent years, primarily involving aerosol sprays.
Why was secret deodorant recalled?
Secret aerosol deodorant was recalled in late 2021 after independent and internal testing revealed the presence of benzene, a known human carcinogen. Procter & Gamble voluntarily recalled specific lots of Secret and Old Spice aerosol sprays to prevent potential long-term health risks to consumers.
What is the largest FDA recall on deodorant?
While the FDA does not officially rank recalls by size, the late 2021 Procter & Gamble recall of Old Spice and Secret aerosol sprays is among the largest. Combined with the 2022 recall of Sure and Brut aerosols, these actions impacted millions of units nationwide.
The 2022 Sure and Brut Aerosol Benzene Recall
In February 2022, TCP HOT Acquisition LLC (doing business as HRB Brands) issued a voluntary nationwide recall of Sure and Brut aerosol antiperspirants. The recall applied to all lot numbers with expiration dates on or before August 2023. Like the Secret recall, this was initiated due to the unexpected presence of benzene in the aerosol propellant.
| Year | Brand(s) | Reason for Recall | Scope |
|---|---|---|---|
| 2025 | Power Stick (AP Deauville) | Manufacturing deviations | 67,000+ cases |
| 2022 | Sure & Brut (HRB Brands) | Benzene contamination | All lots expiring on/before Aug 2023 |
| 2021 | Secret & Old Spice (P&G) | Benzene contamination | Specific aerosol lots |
Health Risks Associated with Defective Deodorants
Using a recalled cosmetic product can expose you to unnecessary health risks. The severity of the risk depends on the reason for the recall.
Understanding Manufacturing Deviations vs. Contamination
A manufacturing deviation means the product was not created according to approved, sanitary, or standardized processes. This can lead to ineffective products, bacterial growth, or improper chemical balances. Contamination, on the other hand, means a foreign, harmful substance (like benzene or heavy metals) was introduced into the product during manufacturing.
The Dangers of Benzene Exposure in Aerosol Products
Benzene is classified as a human carcinogen. Exposure to benzene can occur through inhalation, orally, or through the skin. Prolonged exposure to high levels of benzene has been linked to severe health issues, including:
- Leukemia (cancer of the blood-forming organs)
- Blood disorders like anemia
- Damage to the immune system
Skin Irritation, Chemical Burns, and Other Adverse Reactions
Even without carcinogens, defective deodorants can cause immediate physical harm. Improperly formulated deodorants can lead to severe contact dermatitis, chemical burns in the underarm area, painful rashes, and allergic reactions that require medical treatment.
What to Do If You Own a Recalled Deodorant
If you discover that your deodorant is on the FDA recall list, take immediate action to protect yourself and your family.
How to Safely Dispose of Recalled Cosmetics
Do not throw aerosol cans into fires or incinerators, as they can explode. For roll-on products like Power Stick, simply discard the product in your regular household trash. Do not empty the contents down the drain, as defective chemicals can contaminate local water supplies.
Steps to Claim a Refund from the Manufacturer
Manufacturers are legally obligated to provide refunds or replacements for recalled products. To claim yours:
- Take a clear photo of the product, specifically the lot number and UPC code.
- Visit the manufacturer’s official website and look for the recall refund form.
- Call the customer service number provided in the FDA press release.
- Keep your receipt if you still have it, though it is rarely required for a recall refund.
Reporting Adverse Effects to the FDA MedWatch Program
If you experienced a rash, burn, or illness after using a recalled deodorant, report it to the FDA’s MedWatch Adverse Event Reporting program. This helps the FDA track the severity of the issue and can trigger broader safety investigations.
Legal Rights for Consumers Harmed by Recalled Products
While getting a $5 refund covers the cost of the product, it does not cover the medical bills or suffering caused if the defective deodorant injured you.
When Does a Product Recall Lead to a Lawsuit?
A product recall can lead to a lawsuit if the defective product caused actual, documented physical harm. For example, if a consumer develops severe chemical burns from a poorly formulated roll-on, or is diagnosed with a specific cancer linked to benzene exposure from an aerosol spray, they may have grounds for a product liability claim.
Compensation for Medical Bills and Personal Injury
Victims of defective products can seek compensation for:
- Medical expenses (dermatologist visits, oncology treatments, medications)
- Lost wages due to time off work
- Pain and suffering
- Punitive damages in cases of gross corporate negligence
How a Defective Product Attorney Can Help
Navigating a claim against a massive cosmetics corporation is complex. A defective product attorney can help you gather medical evidence, preserve the defective product for testing, and negotiate a fair settlement. If you have suffered a severe reaction or illness linked to an FDA deodorant recall, consulting with a legal professional is the best way to protect your rights.