Table of contents
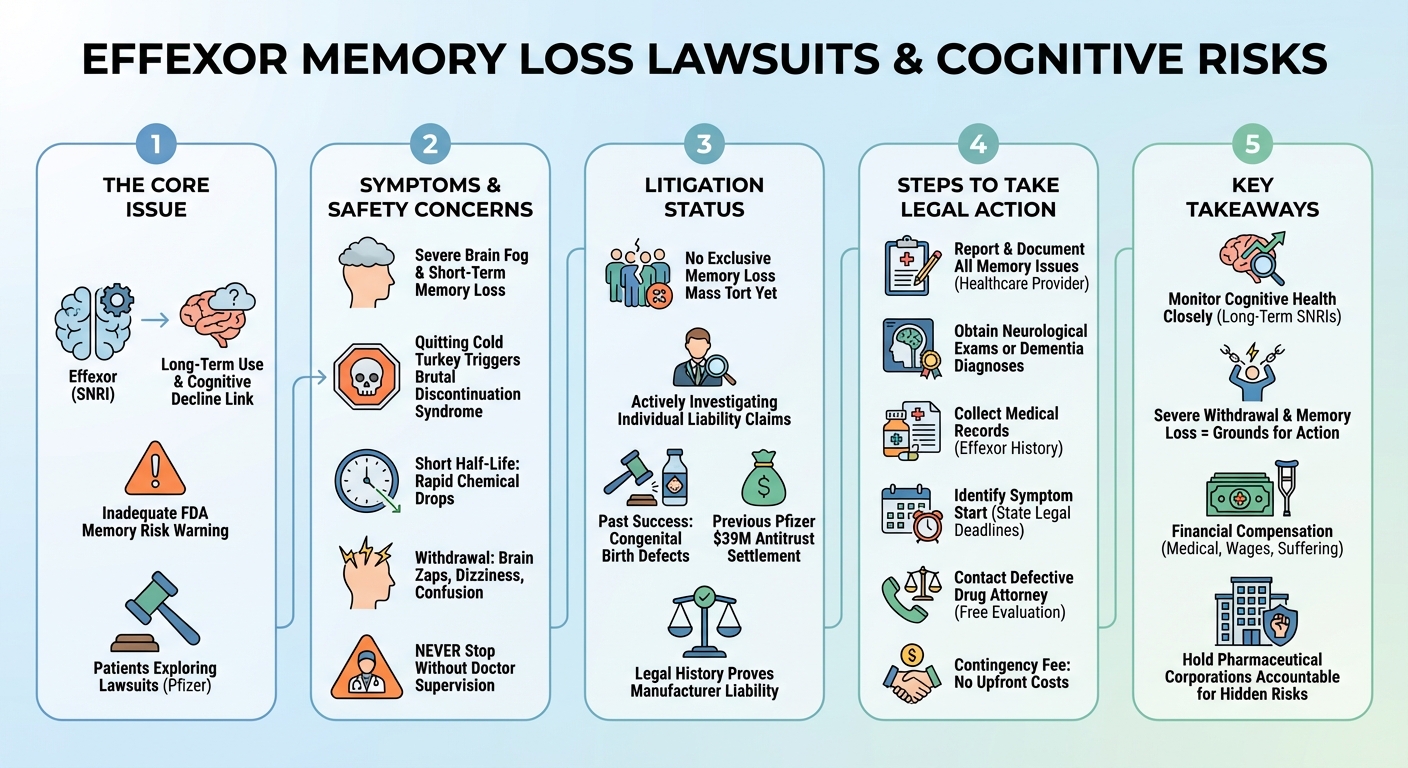
Currently, there is no consolidated class action lawsuit specifically for Effexor-induced memory loss. However, patients experiencing severe cognitive decline, dementia, or extreme discontinuation syndrome after taking venlafaxine are consulting defective drug attorneys. If Pfizer failed to adequately warn about these neurological risks, you may qualify for an individual product liability claim.
Effexor Memory Loss Lawsuits: Current Status and Updates
What is Effexor (Venlafaxine)?
Effexor, known generically as venlafaxine, is a popular prescription medication used primarily to treat major depressive disorder, generalized anxiety disorder, and panic disorder. Manufactured by Wyeth (now a subsidiary of Pfizer), it belongs to a class of drugs called Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs). While effective for many, the drug has been linked to severe side effects, leading to ongoing scrutiny and litigation.
Can you file a lawsuit specifically for memory loss?
Currently, there is no consolidated multidistrict litigation (MDL) or class action lawsuit exclusively dedicated to Effexor-induced memory loss. However, product liability attorneys are actively investigating claims from patients who have suffered severe cognitive decline, early-onset dementia, or permanent memory impairment after taking the drug. If evidence proves that Pfizer knew about these long-term neurological risks and failed to adequately warn the public or healthcare providers, affected patients may have strong grounds for individual defective drug lawsuits.
Overview of active and historical litigation against Pfizer/Wyeth
Pfizer and Wyeth have faced numerous lawsuits over Effexor in the past decade. Historically, litigation focused heavily on congenital birth defects, particularly heart defects in children born to mothers who took the drug during pregnancy. More recently, legal focus has shifted toward the drug’s extreme withdrawal symptoms (discontinuation syndrome) and emerging studies linking long-term SNRI use to cognitive decline. As medical research evolves, so does the potential for new mass torts targeting these undisclosed neurological side effects.
The Link Between Effexor and Cognitive Decline
Does Effexor cause memory issues?
Yes, many patients report short-term memory loss, brain fog, and cognitive impairment while taking Effexor or during withdrawal. Furthermore, recent medical studies are investigating potential links between long-term use of SNRIs like venlafaxine and an increased risk of cognitive decline or early-onset dementia.
How does venlafaxine affect the brain?
Venlafaxine is an SNRI that alters brain chemistry by increasing levels of serotonin and norepinephrine. These neurotransmitters help regulate mood, energy, and focus. However, artificially altering these chemicals can sometimes disrupt normal cognitive processing, leading to side effects like memory consolidation issues and brain fog.
Recent studies on antidepressants and dementia risks
Recent pharmacological studies published in 2024 and 2025 have raised alarms regarding the long-term use of certain antidepressants, including SSRIs and SNRIs. Researchers are finding correlations between prolonged use of these medications and higher rates of cognitive decline and dementia in older adults. While the exact mechanism is still being studied, the potential that these drugs accelerate brain aging or impair memory consolidation is a major concern for patient safety advocates and legal professionals.
Effexor Withdrawal and Discontinuation Syndrome
What happens if you stop Effexor cold turkey?
Stopping Effexor cold turkey triggers a severe condition known as discontinuation syndrome. Patients frequently experience debilitating brain zaps (electrical shock sensations in the head), extreme dizziness, confusion, nausea, and temporary cognitive impairment. You should always taper off this medication slowly under strict medical supervision.
Brain zaps, confusion, and temporary cognitive impairment
The neurological fallout of stopping Effexor can be profound. The hallmark symptom, ‘brain zaps,’ feels like sudden electrical jolts inside the skull, often accompanied by vertigo and disorientation. During this withdrawal phase, patients frequently report severe confusion, an inability to concentrate, and temporary memory loss. For some, these cognitive impairments linger for months after the last dose, severely impacting their ability to work and function normally.
Why Effexor withdrawal is more severe than other antidepressants
Effexor is notorious in the medical community for having one of the most brutal withdrawal profiles of any antidepressant. This is primarily due to venlafaxine’s very short half-life. The drug leaves the bloodstream rapidly, causing a sudden and drastic drop in serotonin and norepinephrine levels. The brain does not have time to adjust to this rapid chemical depletion, resulting in a severe systemic crash that triggers intense physical and cognitive symptoms.
Long-Term Risks and Safety Concerns
Is venlafaxine safe long term?
While the FDA has approved venlafaxine for long-term treatment of depression and anxiety, prolonged use carries notable risks. Long-term safety concerns include potential cardiovascular issues, severe chemical dependency, debilitating withdrawal symptoms upon cessation, and emerging questions regarding cognitive decline and memory impairment after years of use.
Monitoring cognitive health while on SNRIs
Patients prescribed Effexor for extended periods should maintain open communication with their healthcare providers regarding their cognitive health. If you notice persistent brain fog, forgetfulness, or difficulty processing information, it is crucial to document these symptoms. Routine neurological evaluations can help differentiate between symptoms of depression and medication-induced cognitive impairment.
FDA warnings and label updates
The FDA has required several label updates for Effexor over its lifespan. These include strict ‘black box’ warnings regarding the increased risk of suicidal thoughts and behaviors in young adults. The FDA has also mandated warnings about the severity of discontinuation syndrome. However, critics argue that the current warning labels still do not adequately address the risks of long-term memory loss or the potential for permanent cognitive decline.
History of Effexor Litigation: Birth Defects to Antitrust
Congenital heart defects and the 2012-2013 lawsuits
The earliest wave of major Effexor litigation began around 2012, focusing on product liability claims related to birth defects. Families sued Pfizer and Wyeth, alleging that taking Effexor during pregnancy caused severe congenital anomalies, including life-threatening heart defects in newborns. Plaintiffs argued that the manufacturer failed to warn doctors and expectant mothers about these teratogenic risks.
The $39 million antitrust settlement
Beyond personal injury claims, Pfizer also faced legal action regarding its business practices. In a major antitrust class action, direct purchasers accused Wyeth of conspiring with Teva Pharmaceuticals to delay the release of generic venlafaxine, artificially inflating the price of the drug. Pfizer ultimately agreed to a $39 million settlement to resolve these antitrust claims, highlighting the company’s aggressive tactics to protect its profits.
How past settlements impact future product liability claims
While antitrust settlements and birth defect lawsuits involve different legal arguments than memory loss claims, they establish a clear pattern of litigation against the manufacturer. Past cases demonstrate that pharmaceutical giants can be held accountable when they prioritize profits over transparency. This legal history provides a strong foundation for attorneys currently investigating failure-to-warn claims regarding Effexor’s cognitive side effects.
Do You Qualify for an Effexor Lawsuit?
Criteria for a defective drug claim
To qualify for a product liability lawsuit against the manufacturer of Effexor, plaintiffs generally need to prove that the drug was defective (either in design or through a failure to warn) and that this defect directly caused their injuries. For memory loss claims, this means demonstrating that you suffered significant, documented cognitive decline or severe discontinuation syndrome, and that Pfizer failed to adequately warn you or your doctor about these specific risks.
Gathering medical records and evidence of cognitive decline
A successful lawsuit requires robust evidence. If you intend to file a claim, you will need comprehensive medical records showing your history of Effexor prescriptions. Additionally, you must provide medical documentation of your cognitive injuries, such as neurological assessments, memory tests, or diagnoses of dementia or severe cognitive impairment that correlate with your use of the medication.
Statute of limitations for filing your case
Every state has a statute of limitations that restricts the amount of time you have to file a defective drug lawsuit. This timeline typically begins on the date you discovered, or reasonably should have discovered, that your memory loss or cognitive decline was linked to Effexor. Because these deadlines vary widely by state, it is critical to consult with a legal professional immediately to ensure your right to seek compensation is protected.
How Our Defective Drug Attorneys Can Help
Free case evaluations and zero upfront fees
Navigating a complex pharmaceutical lawsuit requires experienced legal representation. Our attorneys offer completely free, confidential case evaluations to help you understand your legal options. We operate on a contingency fee basis, meaning you pay zero upfront costs or out-of-pocket fees. We only get paid if we successfully secure a settlement or verdict on your behalf.
Holding pharmaceutical giants accountable
Pharmaceutical companies have a legal and ethical duty to ensure their products are safe and to warn the public about all potential side effects. When they fail to do so, they must be held financially responsible for the harm they cause. Our legal team has the resources and experience necessary to stand up to massive corporations like Pfizer and fight for the justice you deserve.
Contact us today
If you or a loved one has suffered severe memory loss, cognitive decline, or debilitating withdrawal symptoms after taking Effexor, do not wait to seek help. Contact our defective drug attorneys today to schedule your free consultation and take the first step toward securing the compensation you need for your medical bills, lost wages, and pain and suffering.