Table of contents
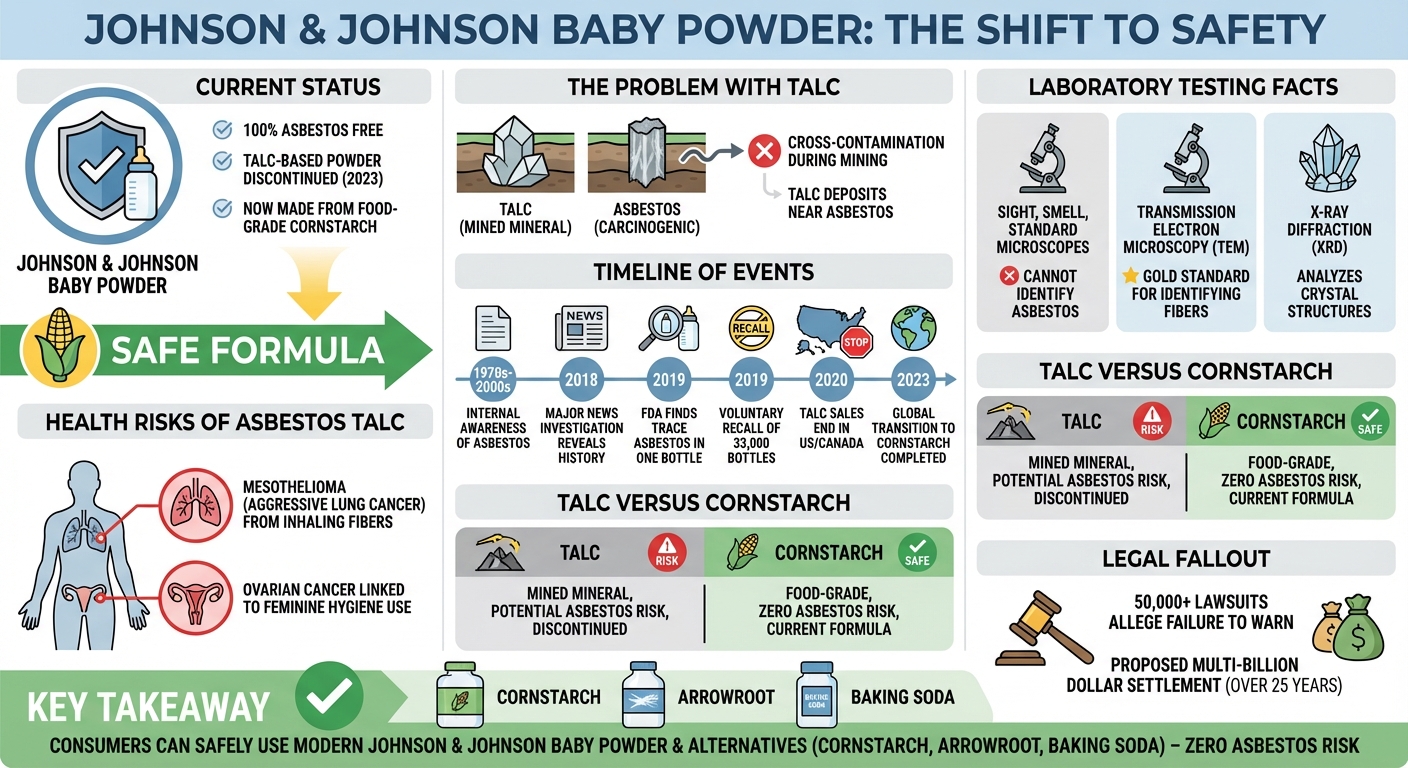
Johnson & Johnson baby powder no longer contains asbestos. Following years of lawsuits and a 2019 FDA recall after trace amounts of asbestos were found in its talc, J&J discontinued its talc-based powder globally. Today, all Johnson’s Baby Powder is made from cornstarch, which is naturally asbestos-free.
The Controversy: Johnson & Johnson Baby Powder and Asbestos
What is the link between talcum powder and asbestos?
Talc is the softest known mineral on earth, widely used in cosmetics for its ability to absorb moisture and reduce friction. However, talc deposits in the earth are frequently located near asbestos, a naturally occurring and highly carcinogenic mineral. During the mining process, it is possible for talc to become cross-contaminated with microscopic asbestos fibers. If not strictly purified and tested, this contaminated talc can end up in consumer products.
Summary of the Reuters investigation and internal J&J documents
In December 2018, a bombshell investigative report by Reuters revealed that Johnson & Johnson executives, mine managers, and doctors were aware of occasional asbestos contamination in their talc supply. According to internal company memos spanning from the early 1970s to the early 2000s, J&J’s raw talc and finished powders sometimes tested positive for small amounts of asbestos. The investigation alleged that the company kept this information from regulators and the public, a claim that Johnson & Johnson has vehemently denied, maintaining that their talc has always been safe.
FDA Testing and the 2019 Product Recall
How the FDA found chrysotile asbestos in J&J talc
In 2019, the U.S. Food and Drug Administration (FDA) conducted routine testing on various cosmetic products for asbestos. During this testing, the FDA discovered trace amounts of chrysotile asbestos—one of the most common types of asbestos—in a single bottle of Johnson’s Baby Powder purchased from an online retailer.
Johnson & Johnson’s response and voluntary recall
Following the FDA’s discovery, Johnson & Johnson initiated a voluntary recall of 33,000 bottles of baby powder from the affected lot in October 2019. The company stated this was done out of an abundance of caution. J&J subsequently conducted its own third-party testing on the same bottle and lot, claiming they found no presence of asbestos and suggesting the FDA’s results may have been compromised by environmental contamination or testing errors.
The science of testing talc for asbestos contamination
Testing talc for asbestos requires highly specialized laboratory equipment. Standard microscopes cannot detect the microscopic fibers. The most reliable methods include:
- Transmission Electron Microscopy (TEM): Uses a beam of electrons to view the atomic structure of fibers, making it the gold standard for identifying asbestos.
- X-Ray Diffraction (XRD): Analyzes the crystal structure of a sample to detect the presence of asbestos minerals.
Health Risks Associated with Asbestos-Contaminated Talc
Mesothelioma and inhalation risks
When asbestos-contaminated powder is applied, microscopic fibers can become airborne and be inhaled. Over decades, these trapped fibers can cause inflammation and genetic scarring in the lungs. This exposure is directly linked to mesothelioma, an aggressive and deadly cancer of the lung lining (pleura) or abdominal cavity.
Ovarian cancer claims and ongoing research
Beyond inhalation risks, tens of thousands of lawsuits allege that using talcum powder for feminine hygiene caused ovarian cancer. Plaintiffs argue that asbestos-laced talc particles can migrate through the reproductive system to the ovaries. While the scientific community is somewhat divided—with some epidemiological studies showing a slight increased risk and others showing no definitive link—the International Agency for Research on Cancer (IARC) classifies the perineal use of talc-based body powder as “possibly carcinogenic to humans.”
Current Product Safety: Is J&J Baby Powder Safe Now?
The global transition from talc to cornstarch
Facing mounting legal pressure and declining sales, Johnson & Johnson discontinued the sale of talc-based baby powder in the United States and Canada in 2020. In 2023, the company expanded this decision globally. Today, all Johnson’s Baby Powder is manufactured using cornstarch.
| Feature | Talc-Based Powder (Discontinued) | Cornstarch-Based Powder (Current) |
|---|---|---|
| Origin | Mined mineral | Food-grade crop derivative |
| Asbestos Risk | Potential for cross-contamination | Zero risk |
| Current Status | Discontinued globally by J&J in 2023 | Currently sold worldwide |
Does Johnson and Johnson baby powder still have asbestos?
No, Johnson & Johnson baby powder no longer contains asbestos. In 2023, the company transitioned its global portfolio entirely to cornstarch-based baby powder. Because cornstarch is a food-grade ingredient derived from crops rather than a mined mineral, it carries zero risk of naturally occurring asbestos contamination.
Which powder is asbestos free?
Any cosmetic or baby powder made from cornstarch, arrowroot, tapioca, or baking soda is naturally asbestos-free. Since 2023, all newly manufactured Johnson’s Baby Powder is exclusively cornstarch-based, making it a safe alternative completely free of both talc and any potential asbestos contamination.
Identifying Asbestos in Consumer Products
How to identify asbestos powder?
You cannot identify asbestos powder by sight, smell, or touch. Because asbestos fibers are microscopic and blend seamlessly into other dust, the only way to definitively identify asbestos contamination in talcum powder is through rigorous laboratory testing using Transmission Electron Microscopy (TEM).
What products still contain asbestos today?
While banned in many consumer cosmetics, asbestos is still legally used in the United States in certain industrial and automotive products. Today, products that may still contain asbestos include aftermarket brake pads, clutches, specialized roofing materials, gaskets, and some imported corrugated cement sheets.
The Legal Fallout: Lawsuits and Settlements
Overview of consumer lawsuits against J&J
Johnson & Johnson faces over 50,000 lawsuits from consumers alleging that the company’s talc-based baby powder caused their mesothelioma or ovarian cancer. Plaintiffs argue the company knew about the asbestos risks for decades but failed to warn consumers, choosing instead to protect their iconic brand.
Bankruptcy filings and proposed settlement updates
To manage the overwhelming litigation, J&J attempted a controversial legal maneuver known as the “Texas Two-Step.” They created a subsidiary, transferred all talc liabilities to it, and immediately filed that subsidiary for Chapter 11 bankruptcy. While courts initially rejected this strategy, J&J continues to pursue settlement frameworks, recently proposing a multi-billion dollar settlement plan intended to resolve all current and future ovarian cancer claims over the next 25 years.