Table of contents
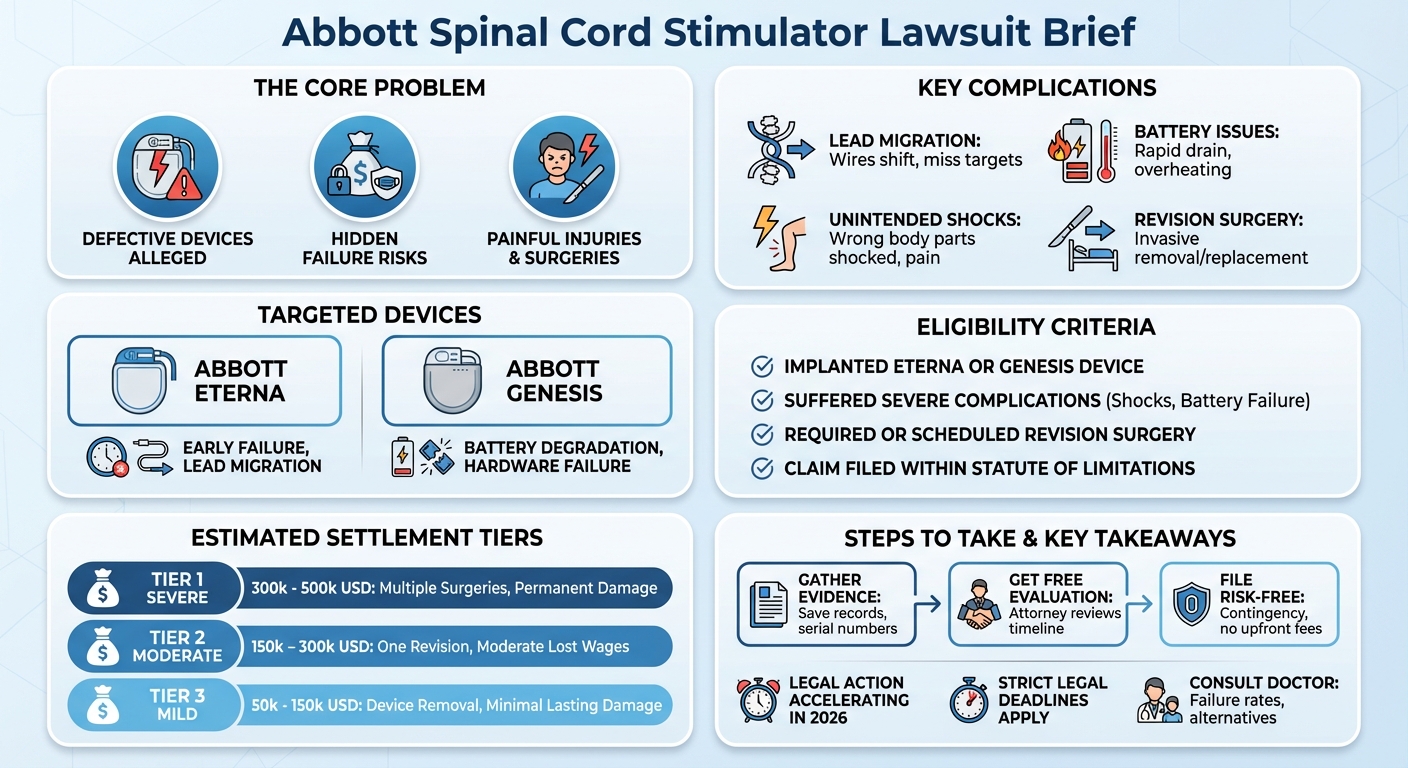
The Abbott spinal cord stimulator lawsuit alleges that defective devices, such as the Abbott Eterna and Genesis systems, cause severe injuries due to lead migration and battery failures. Plaintiffs claim Abbott failed to warn patients about these risks, which often result in painful electrical shocks and require invasive revision surgeries.
Abbott Spinal Cord Stimulator Lawsuit: 2026 Updates and Payouts
Patients who relied on Abbott spinal cord stimulators (SCS) for chronic pain relief are coming forward with claims of severe complications. Recent lawsuits allege that defective Abbott devices have caused unintended electrical shocks, nerve damage, and the need for invasive revision surgeries. If you or a loved one suffered injuries from an Abbott Eterna or Genesis system, you may be entitled to significant financial compensation.
The Rise of Abbott SCS Litigation
Spinal cord stimulators are surgically implanted devices designed to intercept pain signals before they reach the brain. While intended to improve quality of life, a growing number of patients report that Abbott’s devices are failing prematurely. Litigation has steadily increased as more patients discover that their worsening pain or sudden shocks are linked to hardware malfunctions rather than their underlying medical conditions.
Why Patients Are Filing Claims in 2026
In 2026, the legal landscape surrounding Abbott SCS devices has accelerated. Patients are filing claims primarily because they required emergency revision surgeries to remove or replace migrating leads and failing batteries. Many plaintiffs argue that had they known the true risks associated with these specific Abbott models, they would have chosen alternative pain management therapies.
What is the lawsuit against the spinal cord stimulator system?
The lawsuit against the spinal cord stimulator system alleges that manufacturers, including Abbott, produced defective devices that cause severe injuries. Plaintiffs claim the companies failed to warn patients and doctors about risks like lead migration, battery failure, and unintended shocks, often requiring painful revision surgeries to remove the faulty implants.
Core Allegations Against Abbott Laboratories
At the center of the litigation are claims that Abbott Laboratories prioritized profits over patient safety. Lawsuits argue that the company knew or should have known about the high failure rates of their SCS devices but continued to market them as safe and effective.
Failure to Warn About Lead Migration and Device Malfunction
A primary legal argument is the “failure to warn.” Plaintiffs allege that Abbott did not provide adequate warnings to the medical community or patients regarding the likelihood of lead migration—where the electrical wires shift out of place—and subsequent device malfunction. This lack of transparency prevented patients from making informed healthcare decisions.
Specific Devices Involved: Abbott Eterna and Genesis Systems
The lawsuits heavily focus on specific product lines:
- Abbott Eterna: Marketed as the smallest implantable, rechargeable SCS, but recently cited in lawsuits for failing within months of implantation due to lead migration.
- Abbott Genesis: An older system approved in 2001, which has faced long-standing complaints regarding battery degradation and hardware failures.
Latest Updates in the Abbott SCS Litigation (March 2026)
The legal proceedings against Abbott are evolving rapidly. As of March 2026, courts are seeing an influx of product liability claims specifically targeting the Eterna and Genesis models.
Recent Filings: Tuttle v. Abbott and Beyond
Recent high-profile filings, such as Tuttle v. Abbott, highlight the severity of the defects. In this case, the plaintiff alleges that an Abbott Eterna device failed just two months after implantation due to severe lead migration, resulting in debilitating pain and the immediate need for surgical intervention. Similar cases are being filed nationwide.
Current Status of Mass Tort Consolidation
Because of the growing number of similar claims, legal experts are monitoring the potential for multidistrict litigation (MDL) or mass tort consolidation. Consolidating these cases would streamline the discovery process and potentially lead to global settlement negotiations, though individual lawsuits are currently being filed in various federal courts.
What These Updates Mean for New Plaintiffs
For patients who have recently suffered an Abbott SCS failure, these updates are encouraging. The groundwork laid by early plaintiffs makes it easier for new victims to establish a link between the device defects and their injuries. Time is of the essence, however, as statutory deadlines apply.
What are the side effects of Abbott spinal cord stimulator?
The primary side effects of a defective Abbott spinal cord stimulator include severe lead migration, unintended or painful electrical shocks, device overheating, and sudden battery failures. These complications can lead to worsening chronic pain, nerve damage, and the immediate need for invasive revision surgeries to replace or remove the device.
Lead Migration and Unintended Shocks
When the leads (wires) of the stimulator shift from their original placement along the spine, the electrical pulses miss their target. This not only stops the pain relief but can send jolts of electricity into other parts of the body, causing sudden, severe pain and muscle spasms.
Battery Failures and Overheating
Some patients have reported that the implant’s battery drains much faster than advertised or fails to hold a charge. In rare but severe cases, the battery compartment can overheat beneath the skin, causing tissue damage and localized pain.
Severe Pain and the Need for Revision Surgery
The ultimate consequence of these side effects is the necessity of revision surgery. Removing or repositioning an SCS device is an invasive procedure that carries its own risks, including infection, spinal fluid leaks, and extended recovery times. Plaintiffs are seeking compensation for the physical and emotional toll of these additional surgeries.
Eligibility: Who Can File an Abbott SCS Lawsuit?
Not everyone with an Abbott device qualifies for a lawsuit. Your eligibility depends on the specific complications you experienced and the medical interventions required to fix them.
Criteria for a Valid Abbott Claim
To qualify for an Abbott spinal cord stimulator lawsuit, you generally must meet the following criteria:
Statute of Limitations and Filing Deadlines
Every state has a statute of limitations that dictates how long you have to file a product liability lawsuit. This window typically ranges from one to three years from the date you discovered the injury or had the revision surgery. Missing this deadline means losing your right to compensation.
Gathering Your Medical Records and Device Details
Building a strong case requires evidence. Your legal team will need your surgical records, the specific serial and model numbers of your Abbott device, and documentation of your revision surgery. Do not discard any medical paperwork or the explanted device if your surgeon gives it to you.
What is the average settlement for a spinal cord stimulator?
The average settlement for a spinal cord stimulator lawsuit is estimated to range between $100,000 and $500,000, depending on the severity of the injuries. Cases involving multiple revision surgeries, permanent nerve damage, or significant lost wages typically command higher payouts, while less severe complications may settle in lower tiers.
Estimated Compensation Tiers for Abbott Claims
While no two cases are identical, settlement estimates generally fall into tiers based on the extent of the patient’s suffering:
| Injury Severity | Estimated Settlement Range | Typical Case Profile |
|---|---|---|
| Tier 1 (Severe) | $300,000 – $500,000+ | Multiple revision surgeries, permanent nerve damage, permanent disability. |
| Tier 2 (Moderate) | $150,000 – $300,000 | One revision surgery required, prolonged recovery, moderate lost wages. |
| Tier 3 (Mild) | $50,000 – $150,000 | Device failure requiring removal, minimal lasting physical damage. |
Factors Influencing Your Settlement Amount
Your individual payout will be influenced by your age, your occupation, the total cost of your medical bills, and the degree to which the device failure impacted your daily life. Punitive damages may also be awarded if a jury finds Abbott’s conduct to be particularly negligent.
Compensation for Revision Surgeries and Lost Wages
A successful settlement should cover all past and future medical expenses related to the defective device. Furthermore, it should reimburse you for any income lost during your recovery periods and compensate you for the physical pain and emotional distress endured.
Which company has the best spinal cord stimulator?
Determining which company has the best spinal cord stimulator depends on individual patient needs, but Abbott, Medtronic, and Boston Scientific are the top market leaders. While each offers advanced pain management technology, all three companies have faced product liability lawsuits regarding device malfunctions, lead migration, and battery failures.
Comparing Abbott, Medtronic, and Boston Scientific
The “Big Three” in the SCS market all offer unique features. Abbott is known for its BurstDR stimulation and low-profile Eterna battery. Medtronic is recognized for its MRI-compatible devices, and Boston Scientific is praised for its highly customizable programming. However, no device is immune to hardware failures.
Why Market Leaders Still Face Defect Litigation
Despite their market dominance, these companies face litigation because spinal cord stimulation involves placing delicate electronics into a highly mobile and sensitive part of the human body. When companies rush products to market or fail to disclose known failure rates, patients suffer the consequences.
Discussing Alternatives with Your Doctor
If you are considering an SCS or need a replacement, have a candid discussion with your pain management specialist. Ask about the failure rates of specific models, inquire about alternative therapies like targeted drug delivery or physical therapy, and ensure you fully understand the risks of lead migration.
How to Start Your Abbott SCS Legal Claim
Taking legal action against a massive medical device manufacturer can feel daunting, but you do not have to do it alone. Securing experienced legal representation is the first step toward recovering the compensation you deserve.
Schedule a Free Case Evaluation
The process begins with a free, confidential case review. A mass tort attorney will listen to your story, review the timeline of your implantation and revision surgeries, and determine if you meet the criteria to file a lawsuit against Abbott.
No Upfront Fees: Understanding Contingency Representation
Product liability attorneys handle Abbott spinal cord stimulator lawsuits on a contingency fee basis. This means you pay nothing out of pocket to hire a lawyer. Your legal team only gets paid a percentage of your settlement or verdict if they successfully win your case. If you don’t win, you owe nothing.