Table of contents
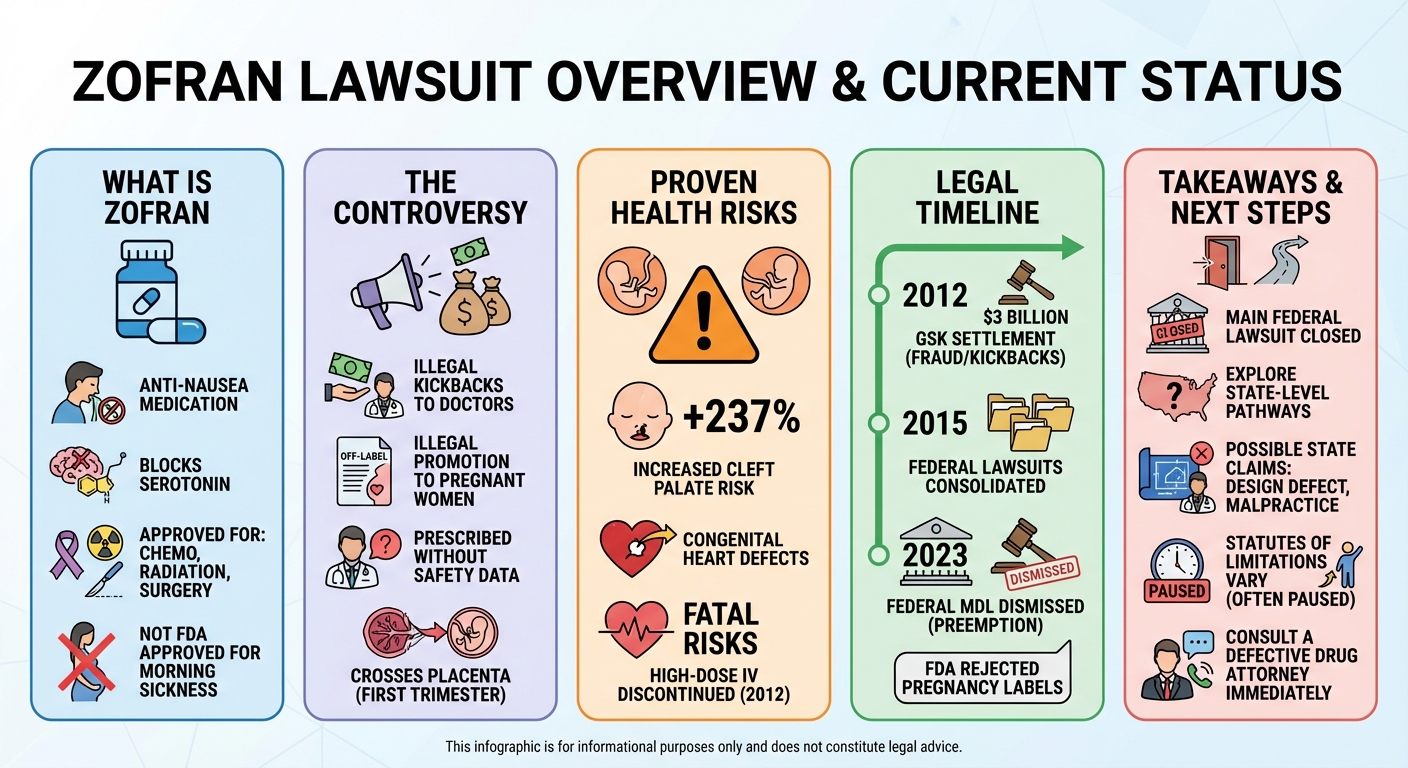
The Zofran lawsuit involves claims against GlaxoSmithKline alleging the company illegally marketed the anti-nausea drug to pregnant women off-label, leading to severe birth defects like cleft palates and heart abnormalities. In 2023, a federal appeals court dismissed the primary multidistrict litigation (MDL) based on federal preemption.
Zofran Lawsuit Updates: Birth Defects, MDL Status, and Legal History
For years, pregnant women suffering from severe morning sickness were prescribed Zofran, a powerful anti-nausea medication. However, allegations soon emerged that the drug’s manufacturer, GlaxoSmithKline (GSK), illegally marketed the drug off-label to expectant mothers. This sparked thousands of lawsuits claiming that Zofran caused severe birth defects, including cleft palates and congenital heart abnormalities. While the legal landscape has shifted significantly in recent years, understanding the history and current status of the Zofran lawsuit is crucial for affected families.
Current Status of the Zofran Lawsuit (2024 Update)
The trajectory of Zofran litigation changed dramatically following key federal court decisions. Families seeking compensation must understand where the multidistrict litigation (MDL) stands today.
The 2023 U.S. Appeals Court Ruling
In January 2023, a U.S. Appeals Court upheld a lower court’s decision to dismiss the Zofran Birth Defects MDL. The centralized litigation, which had consolidated thousands of lawsuits in a Massachusetts federal court, was effectively ended. The court ruled in favor of GSK, determining that the plaintiffs’ claims were barred by federal law.
Understanding Federal Preemption in the Zofran MDL
The dismissal of the MDL was based on a legal doctrine known as “federal preemption.” The judge ruled that state-law claims—specifically that GSK failed to provide adequate warning labels about birth defect risks—were preempted by federal law. Because the FDA had previously reviewed data and rejected requests to add pregnancy warnings to Zofran’s label, the court concluded that GSK could not be sued under state law for failing to include those exact warnings.
What is Zofran and How Was It Used for Morning Sickness?
Zofran is the brand name for ondansetron, a prescription medication originally developed to treat severe nausea.
Off-Label Prescribing Practices
The FDA approved Zofran strictly for patients experiencing nausea and vomiting caused by chemotherapy, radiation therapy, and surgery. It was never FDA-approved for morning sickness. Despite this, it became one of the most widely prescribed off-label treatments for pregnant women experiencing hyperemesis gravidarum (severe pregnancy-related nausea).
How Ondansetron Works
Ondansetron belongs to a class of medications called 5-HT3 receptor antagonists. It works by blocking the action of serotonin, a natural substance in the body that can trigger the nausea and vomiting reflex in the brain and the vagus nerve in the stomach.
Zofran and Birth Defects: Understanding the Risks
The core of the Zofran litigation revolves around the drug’s ability to cross the placenta during the crucial first trimester of pregnancy, potentially interfering with fetal development.
Is Zofran no longer safe in pregnancy?
Zofran is not officially banned for pregnant women, but its safety remains highly controversial. The FDA never approved it for morning sickness. Because several studies link first-trimester use to an increased risk of birth defects, many medical professionals now recommend alternative, FDA-approved treatments for pregnancy-related nausea.
How much does Zofran increase the risk of cleft palate?
Research indicates that taking Zofran during the first trimester can significantly elevate birth defect risks. A prominent Harvard study found that women who took ondansetron early in pregnancy had a 2.37 times higher risk—an increase of roughly 237%—of delivering a baby with a cleft palate compared to those who did not.
Links to Congenital Heart Defects
In addition to orofacial clefts, studies have linked prenatal Zofran exposure to congenital heart defects. The most commonly reported issues include Atrial Septal Defects (ASD) and Ventricular Septal Defects (VSD), which are essentially “holes in the heart” that can require invasive surgeries to repair.
Medical Concerns and FDA Actions
Beyond birth defects, Zofran has faced regulatory scrutiny for other severe health risks.
Why are doctors reluctant to prescribe Zofran?
Doctors are increasingly reluctant to prescribe Zofran to pregnant women because of conflicting studies linking the drug to birth defects like cleft palates and heart murmurs. Additionally, FDA warnings regarding the drug’s potential to cause dangerous heart arrhythmias make physicians cautious about using it off-label.
Why is Zofran discontinued in the US?
While oral Zofran is still available, the FDA forced the discontinuation of the 32 mg single intravenous (IV) dose in 2012. This specific high-dose version was pulled from the U.S. market because it significantly increased the risk of QT prolongation, a potentially fatal heart rhythm disorder.
GlaxoSmithKline (GSK) and the $3 Billion DOJ Settlement
GSK’s marketing practices for Zofran and other drugs led to one of the largest healthcare fraud settlements in U.S. history.
Allegations of Unlawful Promotion
In 2012, the U.S. Department of Justice (DOJ) announced a $3 billion settlement with GSK. A major component of this settlement addressed allegations that GSK unlawfully promoted Zofran for off-label uses, specifically targeting obstetricians and gynecologists to prescribe the drug for morning sickness despite a lack of FDA approval and safety data.
Kickbacks to Physicians
The DOJ also alleged that GSK paid illegal kickbacks to healthcare professionals to encourage them to prescribe Zofran and other medications. These kickbacks reportedly took the form of consulting fees, luxury travel, and expensive meals.
| Legal/Medical Event | Year | Impact |
|---|---|---|
| DOJ Settlement | 2012 | GSK pays $3B for off-label promotion and kickbacks. |
| FDA Warning | 2012 | 32mg IV dose pulled due to heart rhythm risks. |
| MDL Formed | 2015 | Federal lawsuits consolidated in Massachusetts. |
| Appeals Court Ruling | 2023 | MDL dismissed due to federal preemption. |
Can You Still File a Zofran Birth Defect Lawsuit Today?
With the federal MDL closed, the legal pathways for affected families have narrowed, but they are not entirely eliminated.
State vs. Federal Claims
Because the federal mass tort was dismissed based on preemption, plaintiffs cannot currently pursue failure-to-warn claims in federal court. However, depending on specific state laws, some families may still explore individual claims based on different legal theories, such as design defect or medical malpractice if a doctor prescribed the drug negligently.
Statute of Limitations for Birth Injury Cases
The time limit to file a lawsuit, known as the statute of limitations, varies widely by state. In many birth injury cases, the statute of limitations is tolled (paused) until the injured child reaches the age of 18. However, strict “statutes of repose” for product liability can sometimes override this, making prompt legal action essential.
Consulting with a Defective Drug Attorney
If you took Zofran during pregnancy and your child was born with a cleft palate or congenital heart defect, your legal options are complex. It is highly recommended to consult with an experienced defective drug attorney who can evaluate the specifics of your case, determine if any state-level claims remain viable, and guide you through the available legal avenues.